 
Each orbital can be represented by specific blocks on the periodic table. The 1s orbital and 2s orbital both have the characteristics of an s orbital (radial nodes, spherical volume probabilities, can only hold two electrons, etc.) but, as they are found in different energy levels, they occupy different spaces around the nucleus. Orbitals on different energy levels are similar to each other, but they occupy different areas in space. The energy level is determined by the period and the number of electrons is given by the atomic number of the element. The p, d, and f orbitals have different sublevels, thus can hold more electrons.Īs stated, the electron configuration of each element is unique to its position on the periodic table. The four different types of orbitals (s,p,d, and f) have different shapes, and one orbital can hold a maximum of two electrons. Electrons exhibit a negative charge and are found around the nucleus of the atom in electron orbitals, defined as the volume of space in which the electron can be found within 95% probability. Every element on the Periodic Table consists of atoms, which are composed of protons, neutrons, and electrons. The first two subshells of the third shell are filled in order-for example, the electron configuration of aluminum, with 13 electrons, is 1s22s22p63s23p1….2.6: Arrangements of Electrons.\)īefore assigning the electrons of an atom into orbitals, one must become familiar with the basic concepts of electron configurations. The d subshell can hold a maximum of 10 electrons. 
How many electrons are present in an atom in which the 1s 2s 2p 3s and 3p orbitals are filled? The six naturally occurring noble gases are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn)….Noble gas. The portion of Strontium configuration that is equivalent to the noble gas of the preceding period, is abbreviated as. The ground state electronic configuration of Neutral Strontium atom is 5s2. What is the noble gas configuration for strontium atomic number 38? The electron configuration for chlorine is 1s2 2s2 2p6 3s2 3p5. What is the name of element with an electronic configuration of 1s2 2s2 2p6 3s2 3p5? All isotopes of an element have the same electronic structure. The isotope shown here is carbon-12, with a nucleus of 6 protons (red) and 6 neutrons (blue). What is the electronic configuration of carbon 12? Which elements have a noble gas configuration? What is the electronic configuration for carbon? The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og). Noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. Which element has the noble gas configuration? Therefore the C electron configuration will be 1s22s22p2. The remaining two electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for C goes in the 2s orbital. In writing the electron configuration for carbon the first two electrons will go in the 1s orbital. How do you find the electron configuration for carbon? Sodium’s noble gas configuration becomes 3s1. So for sodium, we make the substitution of for the 1s22s22p6 part of the configuration. How do you write a noble gas configuration?Ī noble gas configuration of an atom consists of the elemental symbol of the last noble gas prior to that atom, followed by the configuration of the remaining electrons. List of Noble Gas Configurations for All 118 Elements NUMBER What is the noble gas notation for carbon? What is the noble gas configuration for strontium atomic number 38?. 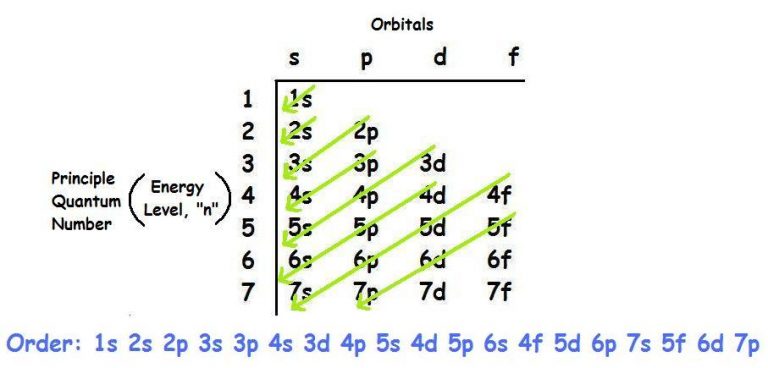
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
